Reconstructing development in a dish to understand how embryos build themselves
Just like bird flocks and snowflakes, post-implantation embryogenesis is an extraordinary example of self-organisation. The emergence of higher-order structures whose complexity exceeds the sum of its parts requires a dynamic dialogue with the extracellular environment and the coordination of cellular behaviours in space and time. Hence, studying embryo development in toto is critical to understand the underlying molecular, cellular and morphogenetic principles regulating this process. While we have gained important insights from tractable and optically accessible species such as zebrafish, important questions, such as how mechanical and chemical inputs are integrated in morphogenesis, remain largely unanswered. Moreover, there is emerging evidence that the regulatory programs and repertoire of cellular behaviours driving morphogenesis differ in mammalian compared to non-mammalian species. For example, axial elongation dynamics differ between mouse and zebrafish, which may be related to differences in e.g. the mechanical forces and the coupling of growth and morphogenesis in mammalian embryos. Such differences illustrate the importance of studying embryo morphogenesis in a mammalian system. In this context, important, yet unanswered questions are: which cell-cell, cell-matrix and tissue-tissue interactions drive mammalian embryo architecture? How are form and fate coordinated? How is developmental variability controlled? In sum, how is a mammalian embryo reproducibly shaped?
In order to fill these knowledge gaps it is useful to investigate self-organised pluripotent-stem-cell-derived stembryos (embryonic organoids). Stembryos circumvent the challenges posed by the inaccessibility of implanted embryos and can be generated in high numbers. Gastruloids are stembryos that form elongating structures reminiscent of an “embryo without a head” with derivatives of all three germ layers, but with limited morphogenesis. For example, somitic cells do not condense into somites and neural cells don’t form a neural tube. We discovered that adding a small percentage of an artificial extra-cellular matrix compound (Matrigel) to gastruloids can unlock their morphogenetic potential. The resulting structures form somites (the precursors of bone, muscle and cartilage in the embryo) and a neural tube (that eventually forms the spinal cord). Given their striking resemblance to the embryonic trunk we called these stembryos "trunk-like-structures" (Fig. 1; Veenvliet et al., 2020; Veenvliet & Herrmann 2021; Bolondi et al., 2021).
Fig. 1: Left: live imaging of the development a trunk-like-structure (TLS). Middle: embryo-like architecture is unlocked in TLS (bottom) compared to gastruloids (top). Right: 3D reconstruction of a TLS carrying fluorescent reporters marking somites (red) and neural tube (cyan), imaged by light-sheet microscopy.
Image credits: Adriano Bolondi, Leah Haut, Dennis Schifferl, Léo Guignard, Jesse Veenvliet.
In contrast to their natural counterpart stembryos are easy to access, track, manipulate and scale, which positions them as a powerful platform to quantitatively interrogate developmental processes across spatial and temporal scales. With Team Stembryo, we will combine these general advantages of stembryos with the unique possibility to directly compare expression and morphogenetic dynamics in stembryos with different levels of morphogenetic complexity (Fig. 2). Through this we will define and dissect the cellular interactions that shape the (st)embryo and show how these impact cell fate decisions. In addition, we will exploit the accessibility and scalability of the system to learn from variation by exploring the stembryo morphospace using multi-modal approaches with the embryo as a "ground truth" reference. We will use the resulting insights to control and steer stembryo development. Together with the implementation of design principles of the embryo, this will result in better, more complete and more reproducible stembryos. Finally, we attempt to generate human trunk-like-structures to enable the study of the molecular, cellular and morphogenetic processes that shape the human post-implantation embryo in health and disease.
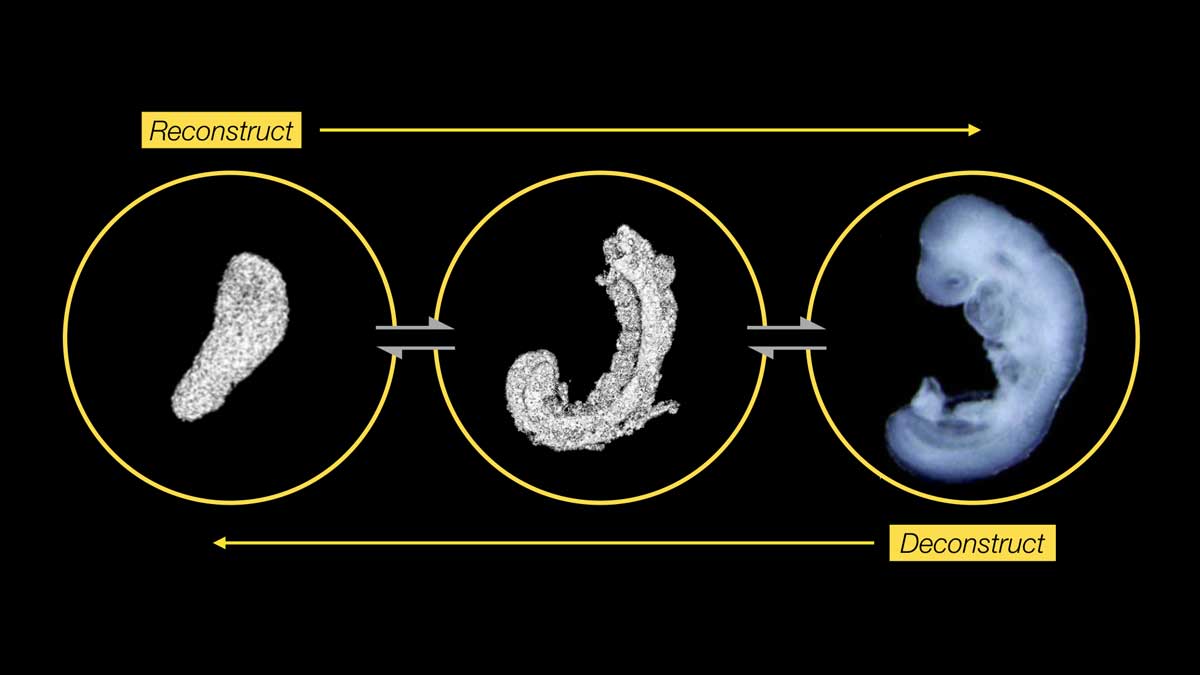
Fig. 2: The morphogenetic potential of gastruloids is unleashed in trunk-like-structures (TLS), resulting in higher-order embryo-like architecture. We will exploit the scalability, tractability and accessibility of stembryos to define the information flows that sculpt the embryo. To this end, we will directly compare molecular, cellular and morphogenetic processes in gastruloids and TLS. Moreover, we will develop tools to learn from variation and utilize the resulting insights to build better, more complete stembryos with higher modularity and reproducibility. Where possible, we include the embryo, the "ground state" truth, in our analyses.