We have developed 2D and 3D (organoid) models of pancreas development in a dish starting from mouse embryonic cells or engineered from human pluripotent stem cells. We study how these systems self-organize. Using genetic modifications we use these models to test how perturbations of genes associated with Diabetes risk predispose to the disease. We have developed image-based high-content screens to upscale our studies.
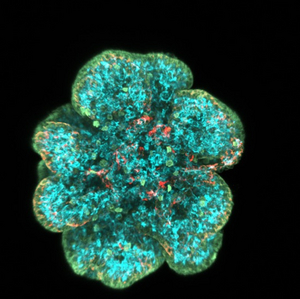
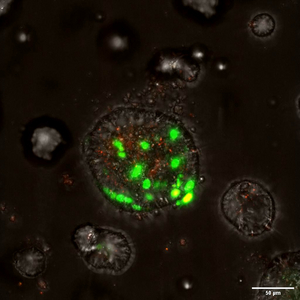
Using human and mouse organoids, 2D stem cell cultures, mouse genetics, and live imaging, we study how each cell makes a decision to proliferate or differentiate, as well as the role of stochastic processes and logic rules. By understanding the processes, we hope to control it and generate numerous beta-cells for diabetes therapy.
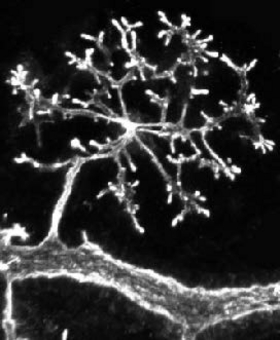
We investigate how the network of ducts and the branching patterns form during pancreas organogenesis. Using organoids, in vivo investigations and modelling in collaboration with mathematicians and physicists, we aim to understand how physical parameters influence morphogenesis and differentiation.
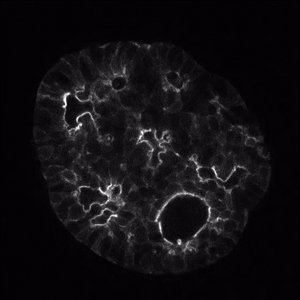
We use human organoids to study how genes expressed during development predispose the fetus to subsequent development of cysts and diabetes.
We investigate how architectural cues deployed along ducts control the differentiation of endocrine cells, notably the links between planar cell polarity, ductal flow and cilia.