Lipids are playing important roles in regulating immune homeostasis. They can be recognized by T cells as antigens when presented by atypical MHC class I proteins of the CD1 family1. The CD1-lipid complex mediated T cell activation is associated with immune disorders and cancer, yet lipid ligands involved are poorly described. Thus, identification of CD1-associated lipids is required to gain further insight into the role of CD1-restricted T cells activation.
CD1 proteins are synthesized in the ER, followed by trafficking through the secretary pathway and endocytic pathway, where cellular lipids in these compartments are loaded or exchanged with foreign lipids. T cells are activated by CD1-lipid complex when it traffics onto cell surface. The analysis of lipids associated with CD1 is technically challenging as detergents required for the extraction of CD1 from membranes leads to the loss of bound lipids. The majority of studies that previously analyzed the spectrum of CD1-associated lipids therefore used genetically modified CD1 proteins that lack the transmembrane domain and that are directly secreted into the cell culture supernatant after passage through the secretory pathway2–4. However, lack of the cytoplasmic tail of CD1 in soluble CD1 proteins is associated with defects in endolysosomal trafficking and thus does not allow the study of lipids that associate with CD1 in these compartments.
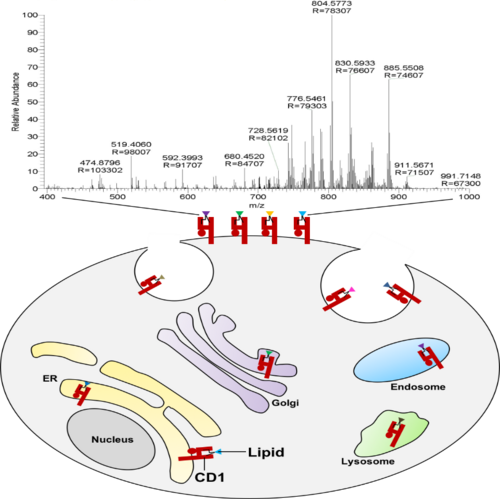
To this end, we developed a cell surface cleavable CD1 protein with intact cytoplasmic tails to study the repertoire of CD1-associated lipids. The engineered CD1 protein carries the recognition motif of a bacterial transpeptidase in its extracellular juxtamembrane domain. The transpeptidase cleaves its recognition sequence, which is not present in eukaryotes, forming an acyl-enzyme intermediate and subsequently links the cleaved protein covalently to an N-terminal triglycine motif 5,6. This strategy leads to a surface cleavable CD1 protein with a C-terminal tag allowing for detergent free extraction and tag-based purification of CD1 that is suitable for the analysis of associated lipids using high resolution mass spectrometry based shotgun lipidomics approach.
In collaboration with:
Prof. Dr. Sebastian Zeissig
The DFG-Center for Regenerative Therapies Dresden (CRTD)
Contact: clinical.lipidomics(at)mpi-cbg.de