During their lifetimes cells must split, repair, and remodel their membrane structures including numerous specialized membrane-bound microenvironments to meet cellular needs. While we know that membrane organization is essential for cellular life, we largely do not understand the underlying molecular mechanisms. This blind spot on our map of the human cell is due the lag of technical approaches able to cover the broad range of scales that molecules bridge to self-organize into membranous organelles. A central question of our research is “How do micron-spanning molecular patterns form and direct molecular activity with atomic precision?”
To address this question, we study how the human cell self-assembles the nucleus, the cells biggest organelle using novel integrated structural biology approaches. Only within a few minutes following every cell division, countless molecules find their place in space and time to reassemble the membrane coated structure that is 10.000 times bigger in diameter than each of its parts. This process – very much like assembling a tent from its tightly packed pieces – provides an example and great case study to dissect the unknown principles leading to Structural Self-Organization of Membranous Organelles. In particular we focus on:
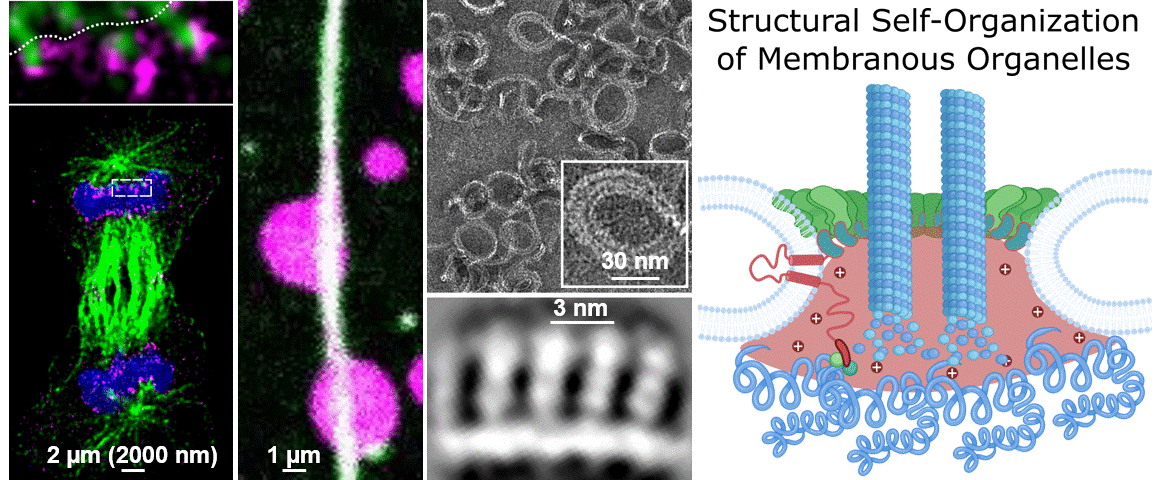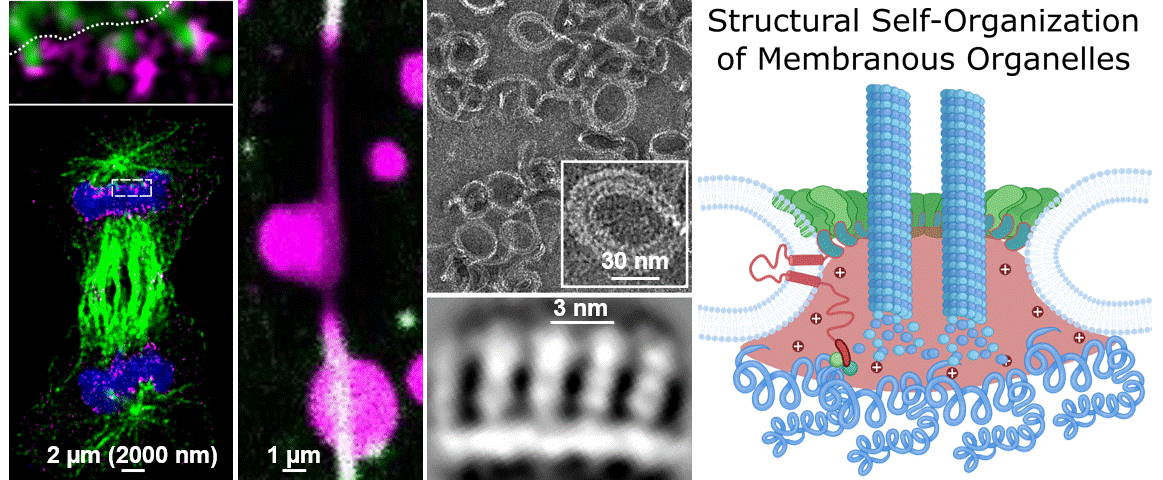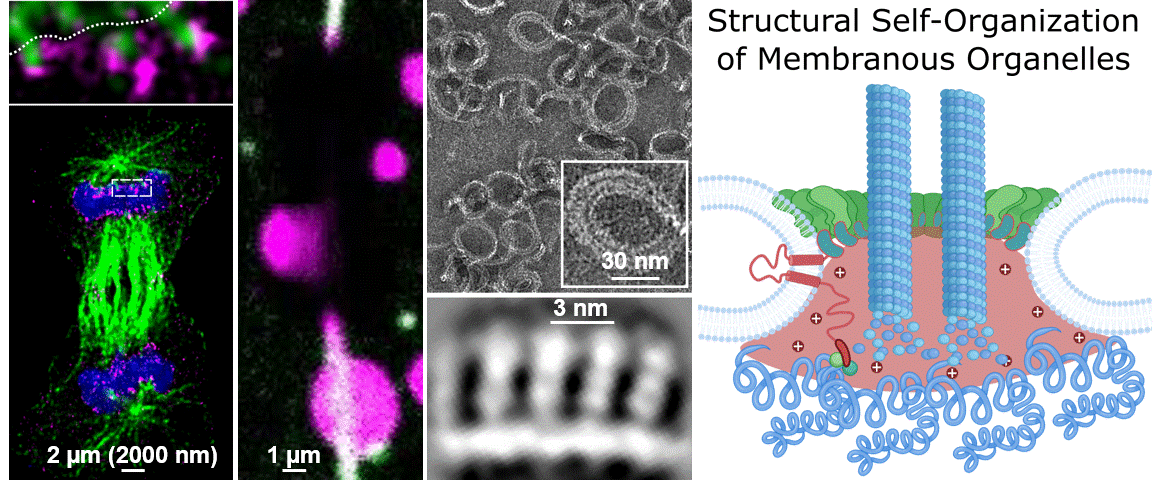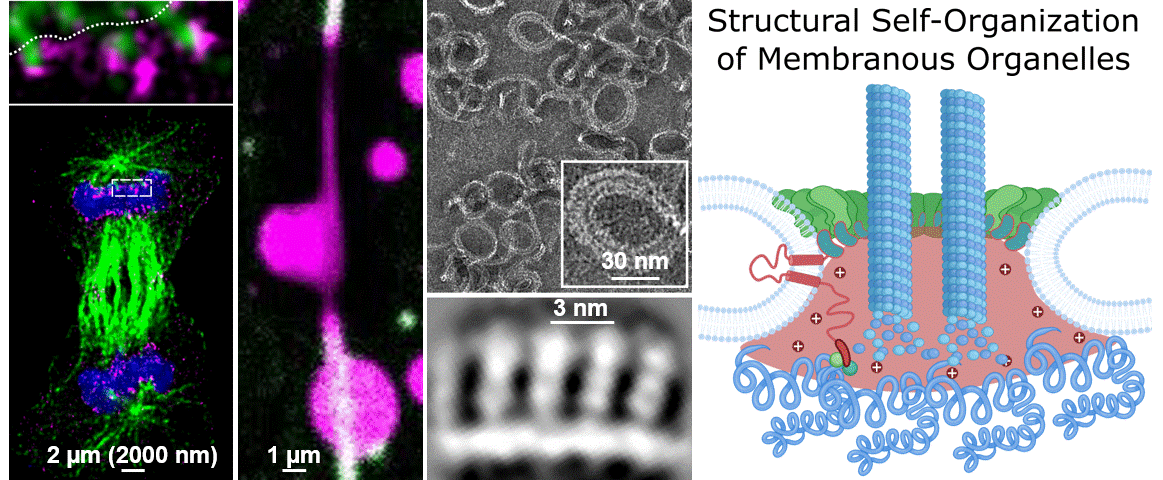
Philosophy: We believe that the best science is the result of a team effort. The world of the unknown is vast compared to our knowledge and it takes creativity to explore it. To do so we have to connect, work together and ignore the boundaries of disciplines. If you are enthusiastic about fundamental biological questions, interested in cutting edge interdisciplinary science, and collaborative, we might be a good match.
For collaborations, internships, PhD and Postdoc position contact: vonappen(at)mpi-cbg.de